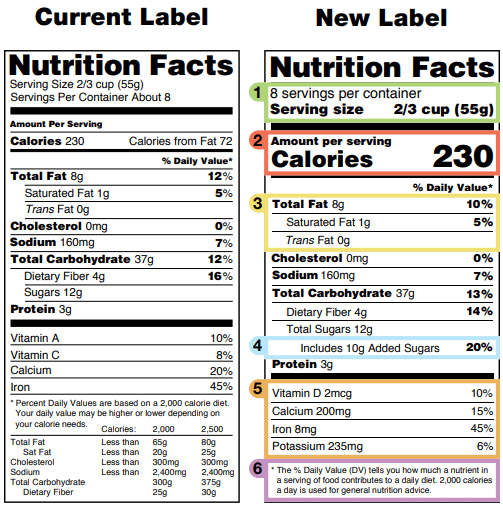
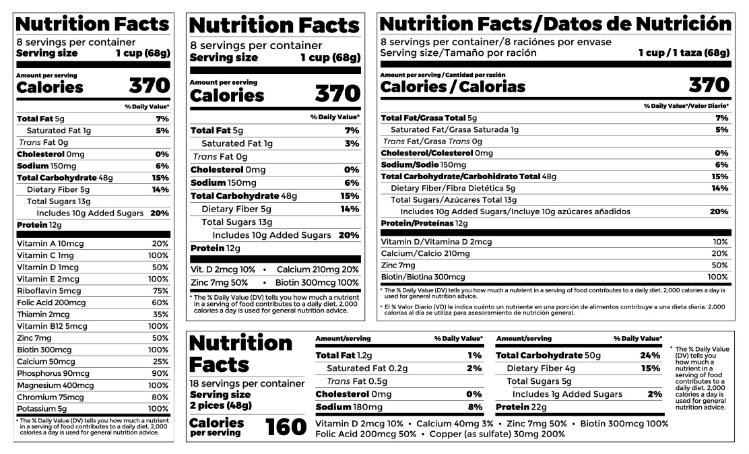
FDA Circular No.2022-005 || Guidelines on Labeling Requirements of Drug Products under Maximum Retail Price (MRP) - Food and Drug Administration

FDA Circular No.2022-005 || Guidelines on Labeling Requirements of Drug Products under Maximum Retail Price (MRP) - Food and Drug Administration