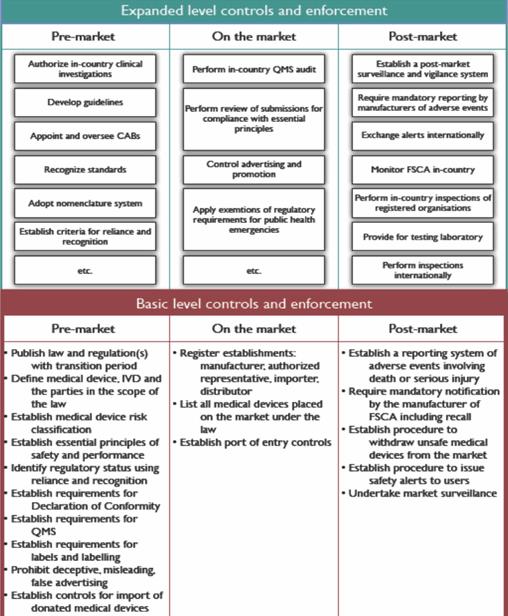
Medical device regulation in Europe – what is changing and how can I become more involved? - EuroIntervention

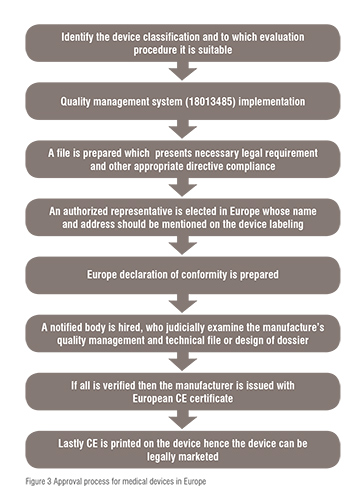
PDF) New Regulations on Notified Bodies and Conformity Assessment of High- Risk Medical Devices in Europe: Impact on Clinical Investigation from an Industry Perspective

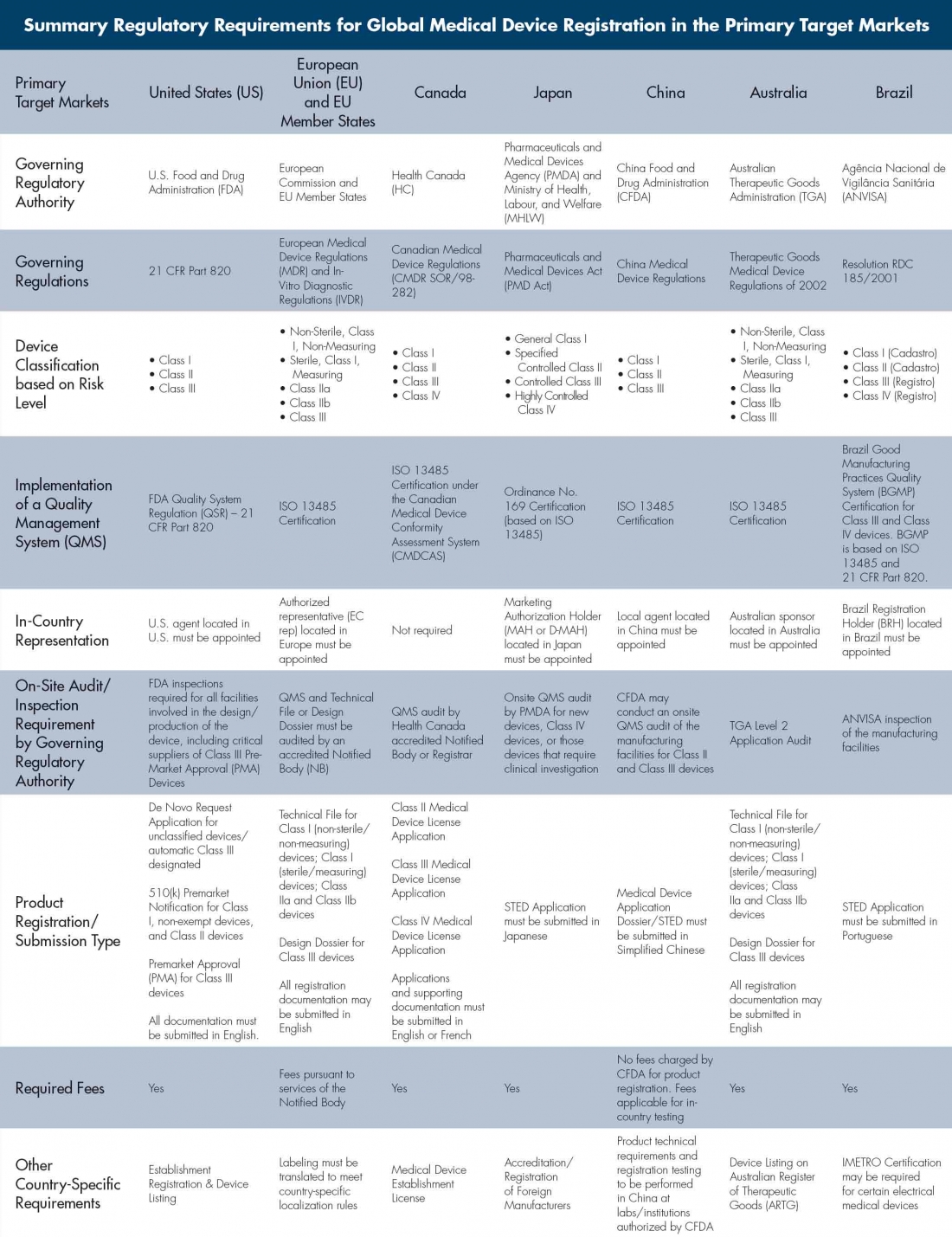
Case Study 28 : Registration of Class II Medical Device in Australia, Brazil, China, India, Pakistan and Russia